Dalton's bold conjecture that all the heavier elements have masses that are integral multiples of hydrogen is not strictly valid, but his theory was a good approximation that eventually led to the discovery of the particles composing the atoms. Magnesium is the eighth most abundant element in Earths crust (about 2.5 percent) and is, after aluminum and iron, the third most plentiful structural metal. 
Notice especially that the atomic mass of hydrogen is not precisely equal to 1, because the atomic mass scale is now based on the most common variety of carbon being exactly 12 atomic mass units. The modern masses for calcium, magnesium, and oxygen are still nearly in the 5:3:2 ratios of the original masses. 
Because the combining mass of oxygen is approximately 16 times that of hydrogen, the preceding chart can be revised, as shown in Table 2. Their masses and abundances are as follows: Table of magnesium isotopes: Atomic Mass Isotopic Abundance 23.98504 u 78.70 24.98584 u 10.13 25.98259 u 11.17. Consequently, he based his scale of atomic masses on hydrogen being equal to 1.Īlthough Dalton's theory was found to be unrealistically simple, he did compel chemists to adopt a standard scale of atomic weights. In 1803, he proposed an atomic theory in which all other elements would be built from multiple hydrogen atoms. Magnesium occurs in nature as a mixture of three isotopes: magnesium-24 (79.0 percent), magnesium-26 (11.0 percent), and magnesium-25 (10.0 percent).Nineteen radioactive isotopes have been prepared magnesium-28 has the longest half-life, at 20.9 hours, and is a beta emitter. The English chemist John Dalton realized that all the known combining masses were nearly whole‐number multiples of the combining mass of the lightest element-hydrogen. 294 72K views 10 years ago A step-by-step explanation of how to draw the Lewis dot structure for Mg (Magnesium). Magnesium compounds were first discovered in a region of Greece known as Magnesia. magnesium and oxygen with a molar mass 40. 
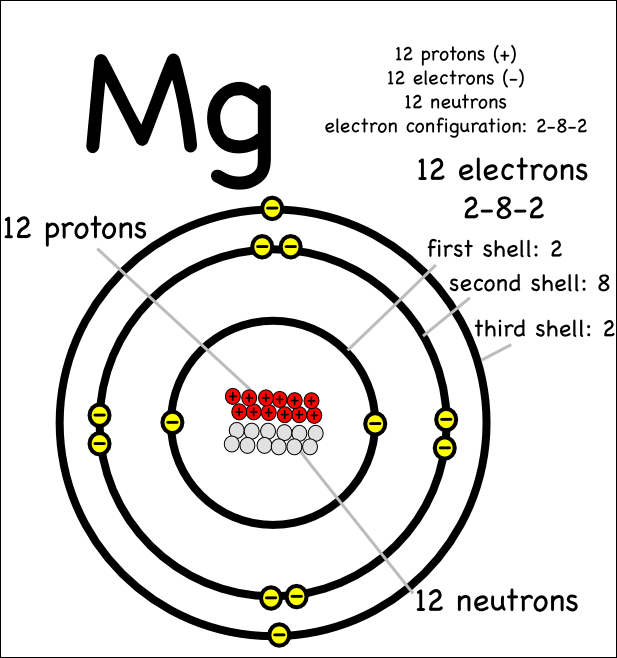
It is the third most abundant element dissolved in seawater, after sodium and chlorine. Its official chemical symbol is Mg, and its atomic number is 12, which means that magnesium has 12 protons in it nucleus. Magnesium is the eighth most abundant element in the Earth's crust and the fourth most common element in the Earth (after iron, oxygen and silicon), making up 13 of the planet's mass and a large fraction of the planet's mantle. Table 1 summarizes these relative combining masses.Ĭhemists gradually discovered that such relative masses in chemical reactions were fundamental characteristics of the elements. Magnesium is a chemical element a substance that contains only one type of atom. The same mass of oxygen, however, required 5 grams of calcium metal to react completely to form calcium oxide. For example, they found that 3 grams of magnesium metal reacted with precisely 2 grams of oxygen to form magnesium oxide with no residual magnesium or oxygen. It was known that differing masses of elements reacted to form compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
